What Is Antibiotic Resistance (AMR) and How Does It Spread?
Share this step
“If we fail to act, we are looking at an almost unthinkable scenario where antibiotics no longer work and we are cast back into the dark ages of medicine.”
-David Cameron, former UK Prime Minister
We have looked at the potential for cutting edge precision medicine to help tackle the rise of diseases such as NCDs; now let’s look at what happens when our existing technologies and solutions are challenged.
Humankind has been battling infection, and the bugs (microbes) that cause it, since ancient Egyptian times. But what we call the “modern era of antibiotics” really started with the discovery of penicillin by Sir Alexander Fleming in 1928. Since then, antibiotics have transformed modern medicine. For example, penicillin was successful in controlling bacterial infections during the second world war, saving millions of lives and making surgery much safer. However, by the 1950s the first cases of antimicrobial resistance (AMR) were documented.
What is antimicrobial resistance (AMR)?
Antimicrobial resistance is when a microorganism (like a bacteria or a virus) can stop an antimicrobial (such as antibiotics, antivirals, and antimalarials) from working against it. Since the 1950s many new antibiotics have been developed, but when these are overused the bugs that they target quickly become resistant rendering the drugs useless.
The first superbug resistant to multiple antibiotics, methicillin-resistant staphylococcus aureus (MRSA), was identified in the U.K. in 1962.
We have now seen resistance to almost every antibiotic we have in use today.
The size and scale of the problem
- Harmful bacteria are living organisms and will fight for survival like every other species.
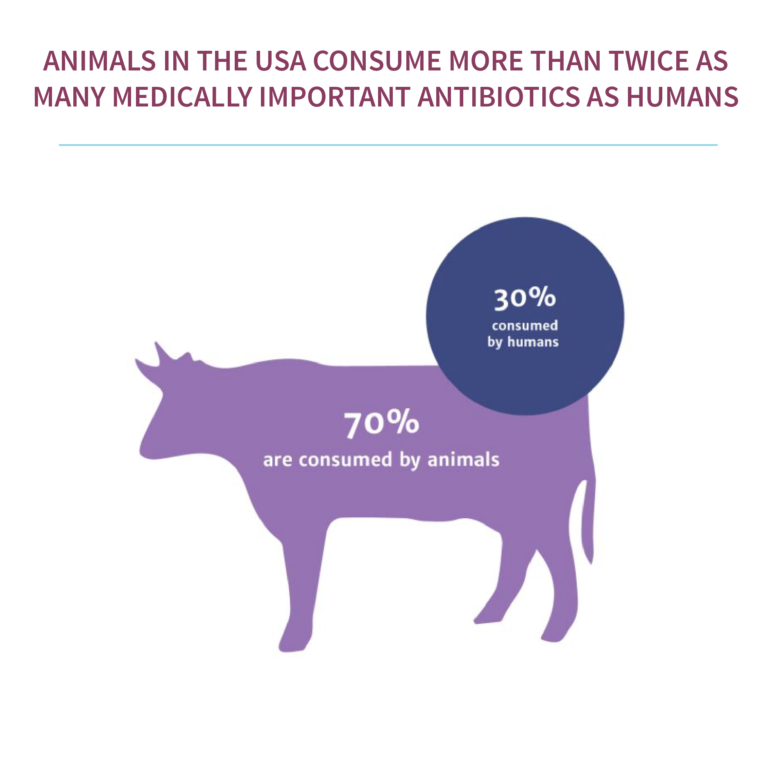
- Antimicrobials leach from animal waste, waste water, and sewage into the natural environment.
How does AMR spread?
- The high use and sometimes misuse of antimicrobial agents.
- Infection of patients with hospital acquired resistant bacteria (e.g., MRSA, Enterobacteriaceiae), which may then be carried back to communities.
- Hospital waste water which releases a high load of antimicrobials and AMR organisms into the environment.
- They can be used indiscriminately, for a partial course of treatment before being discarded.
- They can be given for viral or minor infections where they are not needed.
How can we tackle the problem of AMR?
- Good Infection control and hospital hygiene practice. This includes good levels of staffing, hand hygiene policies, operating theatre procedures, post-operative care, and isolation facilities.
- Generating good community level data. This includes knowing which types of bacteria are in circulation in the community at a given time, and which antibiotics they are susceptible to.
- Education and training of antimicrobial “gate keepers”. This includes training for all antimicrobial prescribers and dispensers, including hospital staff, students, and drug vendors as well as reviewing and updating antimicrobial prescribing guidelines. It also includes having better tools for the recognition of serious and life threatening infections.
AMR and the SDGs
- Globally, at least 700,000 people die each year of drug resistance in illnesses such as bacterial infections, malaria, HIV/AIDS, or tuberculosis and this problem is only set to increase.
- It is estimated that AMR could reduce the global economy by $100 trillion by 2050.
“Strengthen the capacity of all countries, in particular developing countries, for early warning, risk reduction and management of national and global health risks.”
“[W]e will equally accelerate the pace of progress made in fighting malaria, HIV/AIDS, tuberculosis, hepatitis, Ebola and other communicable diseases and epidemics, including by addressing growing antimicrobial resistance and the problem of unattended diseases affecting developing countries.”
Thinking about the other SDGs, we can see how AMR is intertwined with many other global problems.
- SDG 2 which aims to end hunger, will be affected by the use of antibiotics in agriculture.
- SDG 8 which aims to secure productive employment for all.
- SDG 12 which aims to achieve sustainable production and consumption of resources, such as antibiotics.
There is therefore scope within the SDGs for stakeholders working on multiple goals to come together to make tackling AMR part of their agendas. The Global Action Plan on AMR provides a useful starting point. More funding for surveillance (such as this interesting initiative to track the specifics of AMR, and funding for new solutions to the problem (such as through the Longitude Prize) are now needed.
- In your experience, how easy or difficult is it to be prescribed antimicrobials for a common illness such as a cold or flu?
- Do you think that antimicrobials are overused or underused?
Share this

Reach your personal and professional goals
Unlock access to hundreds of expert online courses and degrees from top universities and educators to gain accredited qualifications and professional CV-building certificates.
Join over 18 million learners to launch, switch or build upon your career, all at your own pace, across a wide range of topic areas.
Register to receive updates
-
Create an account to receive our newsletter, course recommendations and promotions.
Register for free

 ©
© 






