Home / History / Archaeology / Archaeology and the Battle of Dunbar 1650: From the Scottish Battlefield to the New World / Isotopes: searching for origins
This article is from the free online
Archaeology and the Battle of Dunbar 1650: From the Scottish Battlefield to the New World


Reach your personal and professional goals
Unlock access to hundreds of expert online courses and degrees from top universities and educators to gain accredited qualifications and professional CV-building certificates.
Join over 18 million learners to launch, switch or build upon your career, all at your own pace, across a wide range of topic areas.

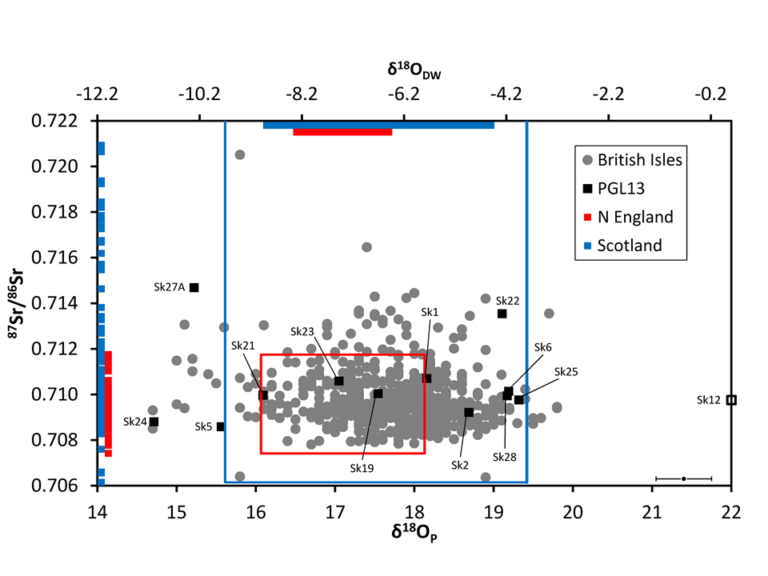 Plot of strontium and oxygen isotope data from Palace Green Library (black) overlaid on 584 published values for individuals buried in Great Britain (Evans et al. 2012) © Andrew Millard, Durham University
Plot of strontium and oxygen isotope data from Palace Green Library (black) overlaid on 584 published values for individuals buried in Great Britain (Evans et al. 2012) © Andrew Millard, Durham University
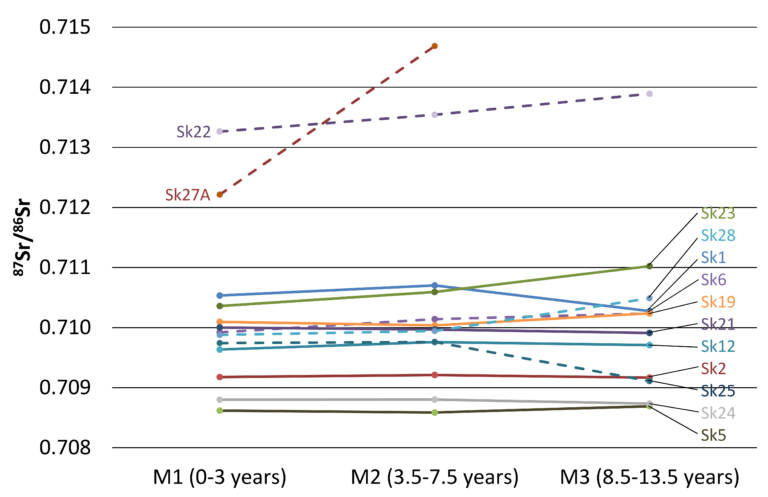 Plot of strontium isotope data on three molars of each man, giving a time series through childhood © Andrew Millard, Durham University
Plot of strontium isotope data on three molars of each man, giving a time series through childhood © Andrew Millard, Durham University






